  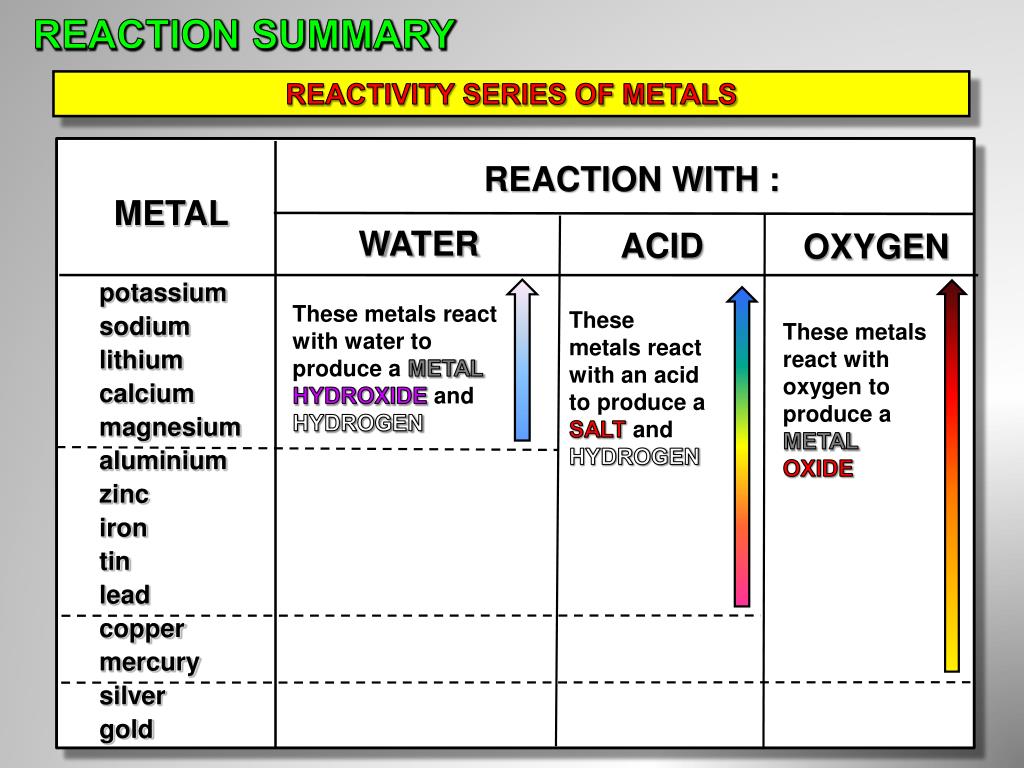
Reactivity is determined by elements’ electron configurations. The chemical stability and comparative reactivity of 207 zinc fingers. Zinc also requires only a single electron to become stable, whereas copper needs two Zinc’s ease of being oxidized makes it much more reactive than copper in many of its chemical reactions. Competition reactions are reactions involving metal and metal oxides The less reactive material, copper, has been displaced from its compound by the more reactive magnesium. Zinc finger reactions with inorganic ions and coordination compounds are as. The order of reactivity is: magnesium > zinc > copper. Ingredients: Zinc oxide HMIS: Health 1, Flammability 0, Reactivity 0 Zinc Oxide. Use the results in the table to deduce an order of reactivity, starting with the most reactive metal. Strong acids, such as hydrochloric or sulfuric acid, can remove the passivating layer and the subsequent reaction with the acid releases hydrogen gas. In an effort to gain a better molecular level understanding of NO and RSNO reactivity with Zn-SR bonds, three families of zinc thiolate model complexes. And zinc plus sulphuric acid gives us zinc sulfate. Extremely pure zinc reacts only slowly at room temperature with acids. Mg(s) + 2HCl(aq) MgCl 2 (aq) + H 2 (g) The more reactive the metal the more rapid the reaction is. Metal displacement: Zn + CuSO 4 ZnSO 4 + Cu Halogen displacement: Cl 2 + 2NaBr 2NaCl + Br 2 Displacement reactions can be observed. Zinc reacts readily with acids, alkalis and other non-metals. Grey magnesium powder and black copper (II) oxide are mixed together, put on some ceramic paper over a tripod with gauze on it For this demonstration goggles or face shields are needed together with safety screens in a well ventilated room The bench should be protected with a large sheet of hardboard In classrooms: one gram of the mixture is strongly heated in a metal crown bottle cap before retiring to the back of the room to await a violent reaction If the reaction doesn't happen, the gas should be turned off and the mixture should be left to cool before being disposed of in water Method: The mixture of powders is heated very strongly At the end, traces of white magnesium oxide are left on the ceramic paper Magnesium + copper(II) oxide -> magnesium oxide + copper Mg (s) + CuO (s) -> MgO (s) + Cu (s) This is an example of a competition reaction as well as a displacement reaction. Zinc Oxide Grade AZO 66L Precaution: Incompat. The more reactive element displaces the less reactive one from its compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
